|
One example of performing work on an object to give it potential energy is the lifting of a body against the gravitational force of the Earth. 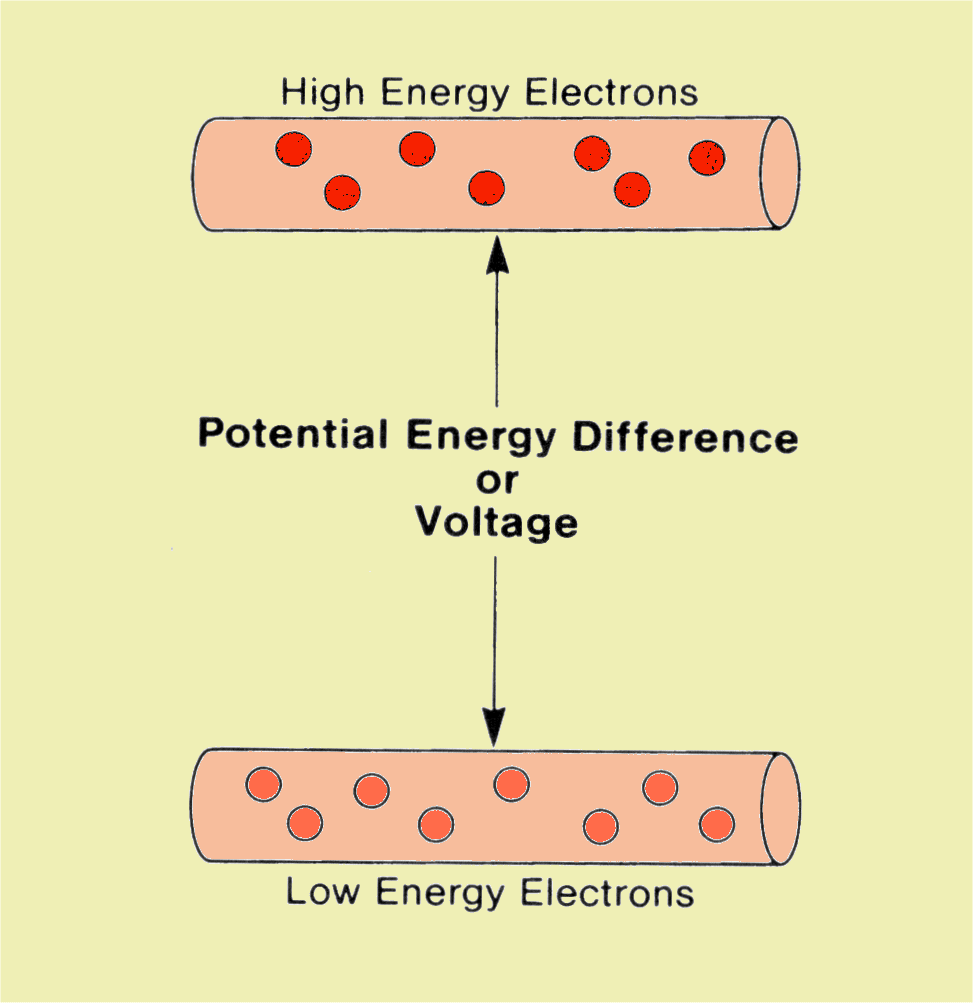
At some point, that object had work performed on it, which resulted in energy storage. An object that has potential energy has energy by virtue of position and is related by the Newtonian equation PE=mgh (where m equals mass in Kg, g the acceleration due to gravity -approximately 9.8 m/s 2 near Earth's surface -and h equals height in meters). Examples of kinetic energy include mechanical energy (caused by motion of parts) and thermal energy (caused by the random motion of particles of matter). The colliding objects can be a hammer swinging down on a nail, or two atoms colliding in a chemical reaction. An object in motion can cause another object to do work by colliding with it, causing it to move a particular distance. Kinetic energy is the energy of an object in motion and is related by the Newtonian formula 1/2mv 2 (where m equals mass in Kg and v equals velocity in meters/second). The Calorie (Cal also called the kilocalorie) that is used to express the energy in food, is equal to 1,000 calories. Energy is often expressed as the calorie (cal), which is the amount of heat needed to raise the temperature of one gram of water by one degree Celsius at a pressure of one atmosphere. The joule is also equal to 1/4.184 of a calorie. 
The Joule is the fundamental unit of energy for both work and heat and is the work done by a force of one Newton acting through a distance of one meter. The Syst ème International d'Unit és (SI) unit for energy is the joule (J), named after James Joule, who demonstrated that work can be converted into heat. Energy transformation is also called energy conversion. The fundamental law of thermodynamics states that the total energy of the universe is fixed and that energy can not be created or destroyed -only converted from one form to another.Įnergy can be changed, or transformed, from one form into another. 
There are many forms of energy (e.g., radiant energy, kinetic energy, potential energy, etc) each of which can be converted into other forms of energy. But the image reception occur with other source: the photossensitivity of many microLEDs with special features of transform photonic energy into voltage (potential energy diference), and that send to the microprocessor to calculate the spatial position of each microreceptiveLED, its voltage intensity to bright.Energy is a state function that is best defined as the capacity to do work or to produce heat. So, the machines uses that energy transforming into its uses (mechanical movement of the obturator, the digital LCD that guide you to take a best picture, etc. Many persons don't fit on this: electric energy is not the energy of the electron: is the kinetic energy that is transformed from waterfalls (hydrelectric), thermoelectric generators, etc, that put potential energy and the potential energy difference (Volt) to electron movements. But the internal energies of the electrons are not used (yet). The electrons are only the carriers of the energy. 
In the camera, as well in many machines you don't use the electron energy, but only the energy that pushes the electrons on the pathway.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
